Online Database of Chemicals from Around the World
| Wuhan Sun-shine Bio-technology Corporation Limited | China | |||
|---|---|---|---|---|
 | www.sun-shinechem.com | |||
 | +86 (27) 6552-2453 | |||
 | +86 (27) 6552-2452 | |||
 | marketing@sun-shinechem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2010 | ||||
| E-fine Bio Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.efine-bio.com | |||
 | +86 15251778053 | |||
 | William.efine@hotmail.com | |||
| Chemical manufacturer since 2021 | ||||
| chemBlink Standard supplier since 2022 | ||||
| Pure Bio Medical Jiangsu Ltd. | China | |||
|---|---|---|---|---|
 | www.purejs.cn | |||
 | +86 (025) 8560-7383 | |||
 | jiangziwei@purejs.cn | |||
| Chemical manufacturer since 2019 | ||||
| chemBlink Standard supplier since 2026 | ||||
| Classification | API >> Other chemicals |
|---|---|
| Name | Pemafibrate |
| Synonyms | (R)-2-[3-[[N-(Benzoxazol-2-yl)-N-[3-(4-methoxyphenoxy)propyl]amino]methyl]phenoxy]butanoic acid; (R)-K 13675 |
| Molecular Structure | 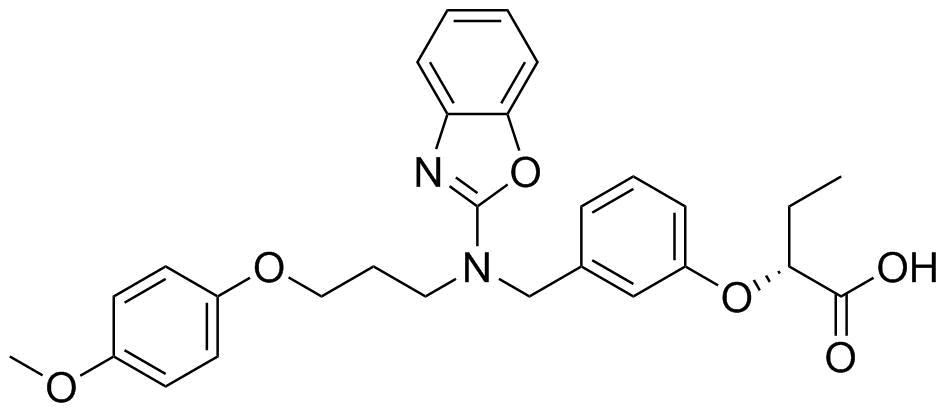 |
| Molecular Formula | C28H30N2O6 |
| Molecular Weight | 490.55 |
| CAS Registry Number | 848259-27-8 |
| SMILES | CC[C@H](C(=O)O)OC1=CC=CC(=C1)CN(CCCOC2=CC=C(C=C2)OC)C3=NC4=CC=CC=C4O3 |
| Solubility | Insoluble (4.4E-4 g/L) (25 $degree$C), Calc.* |
|---|---|
| Density | 1.250$+/-$0.06 g/cm3 (20 $degree$C 760 Torr), Calc.* |
| Melting point | 98-99 $degree$C (ethyl acetate heptane )** |
| Boiling point | 676.7$+/-$65.0 $degree$C 760 mmHg (Calc.)* |
| Flash point | 363.1$+/-$34.3 $degree$C (Calc.)* |
| Solubility | water: freely soluble (Expl.) |
| Index of refraction | 1.619 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software V11.02 ($copyright$1994-2016 ACD/Labs) |
| ** | Yamazaki, Yukiyoshi$semicolon$ Synthesis 2008, (7), P1017-1022. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302-H315-H319-H335 Details |
| Safety Statements | P261-P305+P351+P338 Details |
| SDS | Available |
|
Pemafibrate is a synthetic lipid-lowering drug belonging to the class of selective peroxisome proliferator-activated receptor alpha (PPARα) modulators. It is used primarily for the treatment of dyslipidemia, particularly in patients with elevated triglycerides and abnormal lipid profiles associated with metabolic disorders. The molecular structure of pemafibrate is based on a highly substituted aromatic framework containing multiple functional groups designed to achieve strong and selective binding to the PPARα nuclear receptor. Unlike traditional fibrates, which act as relatively broad PPARα agonists, pemafibrate was developed to provide higher receptor selectivity and improved pharmacological specificity. At the molecular level, pemafibrate binds to the ligand-binding domain of PPARα, a nuclear receptor that regulates the transcription of genes involved in lipid metabolism. Activation of PPARα leads to increased expression of genes responsible for fatty acid β-oxidation, lipoprotein lipase activity, and apolipoprotein production. These effects collectively result in reduced plasma triglyceride levels and modulation of high-density lipoprotein (HDL) cholesterol. Pemafibrate’s structure differs from classical fibrates such as fenofibrate in that it is not structurally based on the fibric acid core. Instead, it contains a more rigid, polycyclic aromatic system with strategically placed polar functional groups that enhance binding affinity and receptor selectivity. This structural optimization allows it to act as a selective PPARα modulator (SPPARMα), a subclass designed to maximize therapeutic effects while minimizing off-target activity. The development of pemafibrate was driven by the need to improve the safety and efficacy profile of fibrate-class drugs. Traditional fibrates can activate multiple PPAR isoforms and are associated with dose-limiting side effects such as liver enzyme elevation or renal effects in some patients. Pemafibrate was designed through structure–activity relationship studies to achieve stronger PPARα activation with reduced interaction with PPARγ and PPARδ. Upon oral administration, pemafibrate is absorbed and distributed systemically, where it enters hepatocytes and binds to nuclear PPARα receptors. The drug–receptor complex heterodimerizes with retinoid X receptor (RXR) and binds to specific peroxisome proliferator response elements (PPREs) in DNA. This regulates transcription of genes involved in lipid catabolism, leading to decreased very-low-density lipoprotein (VLDL) production and enhanced clearance of triglyceride-rich lipoproteins. Clinically, pemafibrate is used to treat hypertriglyceridemia and mixed dyslipidemia, particularly in patients at risk of cardiovascular disease or with metabolic syndrome. It has been investigated as an alternative to conventional fibrates due to its improved selectivity profile and potentially lower risk of adverse effects. Pharmacokinetically, pemafibrate is metabolized primarily in the liver and excreted via biliary pathways. Its chemical stability and receptor selectivity contribute to its once- or twice-daily dosing regimens, depending on clinical indication and formulation. From a medicinal chemistry perspective, pemafibrate represents a new generation of PPARα-targeted agents. Its design incorporates rigid aromatic scaffolds and optimized hydrogen-bonding interactions to enhance receptor binding affinity and selectivity. This distinguishes it from earlier fibrates, which typically rely on simpler aliphatic carboxylic acid structures. Overall, pemafibrate is a selective PPARα modulator used in the management of dyslipidemia. It works by regulating gene expression involved in lipid metabolism, leading to reduced triglyceride levels and improved lipid profiles. Its development reflects advances in nuclear receptor pharmacology aimed at improving efficacy and safety within the fibrate class of drugs. References 2026. Choosing the Right Non-Statin Therapy for the Right Patient – How To Sequence Advanced Lipid-Lowering Therapies. Current Atherosclerosis Reports. URL: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC12945982 2026. Prevalence of substances with OATP1B1 inhibitory properties in individual case safety reports of suspected statin-associated myopathy – an analysis of Swiss pharmacovigilance data. European Journal of Clinical Pharmacology. URL: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC12929307 2007. Design and synthesis of highly potent and selective human peroxisome proliferator-activated receptor alpha agonists. Bioorganic & Medicinal Chemistry Letters. DOI: 10.1016/j.bmcl.2007.05.066 |
| Market Analysis Reports |