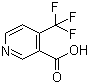
4-(Trifluoromethyl)nicotinic acid, a derivative of nicotinic acid (vitamin B3), features a trifluoromethyl group (-CF3) attached to the 4-position of the pyridine ring. Its chemical formula is C7H4F3NO2, and it is a fluorinated organic compound that has drawn attention in various fields due to its unique structure and reactivity. The introduction of the trifluoromethyl group significantly alters the physicochemical properties of the parent nicotinic acid, imparting increased lipophilicity, thermal stability, and reactivity in certain reactions, which opens up various synthetic and application possibilities.
The discovery of 4-(trifluoromethyl)nicotinic acid can be traced back to research in the field of fluorine chemistry, which has expanded significantly due to the versatile applications of fluorine-substituted compounds in medicinal chemistry and materials science. The trifluoromethyl group is well-known for its ability to influence the biological activity of molecules, making 4-(trifluoromethyl)nicotinic acid an interesting compound for study. Researchers have focused on the synthesis of such compounds due to their potential to interact with biological targets and participate in various organic transformations. Fluorinated nicotinic acid derivatives have become particularly important in the development of pharmaceuticals, where fluorine substitution is used to optimize properties such as metabolic stability, bioavailability, and selectivity.
The synthesis of 4-(trifluoromethyl)nicotinic acid typically involves the introduction of the trifluoromethyl group onto the nicotinic acid backbone through various electrophilic fluorination methods. One common method is the use of trifluoromethylating reagents, such as trifluoromethyl iodide or trifluoromethylsulfonic acid derivatives, which react with the nicotinic acid or its derivatives in the presence of suitable catalysts, typically a transition metal or a strong acid. The synthesis process is usually straightforward, with the trifluoromethyl group being introduced with high efficiency, especially under controlled reaction conditions that minimize side reactions.
In medicinal chemistry, 4-(trifluoromethyl)nicotinic acid has attracted attention for its potential as a pharmacophore in drug discovery. The incorporation of the trifluoromethyl group can enhance the binding affinity of the molecule to certain biological targets, particularly in the modulation of receptors and enzymes involved in neurological or metabolic disorders. For example, nicotinic acid derivatives are known for their influence on the nicotinic acetylcholine receptor, which plays a role in the central nervous system. The trifluoromethyl group may further affect the receptor's binding properties and provide a way to fine-tune the pharmacological profile of nicotinic acid derivatives.
Additionally, the ability of fluorine atoms to alter the electronic properties of the molecule has made 4-(trifluoromethyl)nicotinic acid a useful intermediate in the synthesis of other fluorinated compounds. In the development of agrochemicals, for example, the introduction of trifluoromethyl groups has been shown to improve the activity and stability of certain pesticides. The trifluoromethyl group enhances the lipophilicity of the compound, improving its ability to penetrate biological membranes and interact with target sites. This has made fluorinated nicotinic acid derivatives, including 4-(trifluoromethyl)nicotinic acid, a focus of research in the context of designing new agrochemical agents.
4-(Trifluoromethyl)nicotinic acid has also been explored in the context of material science. The trifluoromethyl group’s electron-withdrawing nature and its effects on molecular polarity have made such compounds useful in the development of organic semiconductors and conducting polymers. These materials, which can be used in electronic devices such as organic light-emitting diodes (OLEDs), organic solar cells, and field-effect transistors, benefit from the unique properties imparted by the trifluoromethyl group. By modifying the electronic properties of the nicotinic acid derivative, researchers can design materials with enhanced performance for use in a range of electronic applications.
Despite the promising applications of 4-(trifluoromethyl)nicotinic acid, challenges remain in its synthesis and application. The presence of the trifluoromethyl group can complicate certain chemical reactions due to the group’s strong electron-withdrawing nature, making some reactions more sluggish or requiring more stringent conditions. Additionally, the environmental impact and toxicity of fluorinated compounds are always a concern in large-scale applications, particularly in the pharmaceutical and agrochemical industries. Further research into the synthesis, biological activity, and environmental effects of such compounds is essential for maximizing their potential while mitigating risks.
In conclusion, 4-(trifluoromethyl)nicotinic acid is a versatile and valuable compound with significant potential in various fields, including medicinal chemistry, agrochemicals, and materials science. Its unique chemical properties, particularly the presence of the trifluoromethyl group, make it an important building block in the design of new pharmaceuticals, pesticides, and advanced materials.
References
2011. Simultaneous determination of flonicamid and its metabolites in vegetables using QuEChERS and reverse-phase liquid chromatography-tandem mass spectrometry. Journal of chromatography. A.
URL: https://pubmed.ncbi.nlm.nih.gov/21831390
|





























































































 GHS07 Warning Details
GHS07 Warning Details